About Lehe
乐于心,和与众,与己乐,与人和; 心宽念纯,百病无生。

Vaccines typically refer to the introduction of antigenic substances into the body, activating the immune system to achieve immune clearance of the pathogen, thereby protecting human health. Vaccines are powerful weapons for humans to actively combat diseases, and can be divided into two major categories: preventive vaccines and therapeutic vaccines based on their application purposes.
Vaccines for infectious diseases are typical representatives of preventive vaccines. Over 200 years ago, Edward Jenner, a British country doctor, used cowpox vaccination to prevent smallpox, pioneering an intervention model of vaccination, pathogen elimination, and human health protection. Vaccination enables the human body to produce long-term immune protection against corresponding pathogens. The World Health Organization (WHO) eradicated smallpox in 1980 through the global implementation of cowpox vaccination. Currently, China implements planned immunization for all newborn infants to prevent infectious diseases such as polio, diphtheria, pertussis, tetanus, measles, and tuberculosis. It can be seen that vaccination technology has become relatively mature and is the most economical and effective means for preventing and controlling infectious diseases.
Cancer vaccines are immunotherapeutic agents based on prepared tumor antigens, which activate the patient's own immune system to induce specific immune responses against tumor cells, ultimately achieving recognition and killing of tumor cells. There are various types of cancer vaccine antigens, including universal tumor-associated antigen vaccines targeting abnormal tumor expression (such as carcinoembryonic antigen CEA vaccines) and whole tumor cell vaccines using intact tumor cells as the antigen source. Among them, personalized neoantigen vaccines prepared by detecting tumor mutations in patients through next-generation gene sequencing (NGS) have attracted particular attention. Research has shown that personalized neoantigen cancer vaccines (protein peptides or mRNA type) can induce a potent and durable specific anti-tumor Tcell response in melanoma, pancreatic cancer, and other tumor patients. Currently, multiple personalized neoantigen cancer vaccines have entered phase II/III clinical studies, demonstrating promising prospects for tumor treatment applications.
For the human body, Tcell-mediated immune protection is one of the most important anti-tumor immune functions. Can cancer vaccines induce a durable anti-tumor Tcell immune protection effect lasting up to 6years in the human body?
▶▶How was the study conducted?
A study published in 2025 in Nature by German researchersexplored the application effect of individualized mRNA vaccines in adjuvant therapy for triple-negative breast cancer (Individualized mRNA vaccines evoke durable T cell immunity in adjuvant TNBC,Nature,2025) and provided an answer to this question.

Triple-negative breast cancer (TNBC) is a highly heterogeneous and aggressive breast cancer subtype, in which the estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) are all negative, accounting for approximately 15% to 20% of all breast cancers. Although there are currently treatment options for TNBC such as the combination of PD-1/PD-L1 inhibitors and chemotherapy, as well as precision therapy with PARP inhibitors for patients with BRCA mutations, this subtype remains a challenge in breast cancer treatment due to its strong aggressiveness, rapid proliferation, high risk of early distant metastasis, and lack of clear targetable markers. However, TNBC exhibits high genomic instability and can express a large number of mutant proteins within tumor cells, presenting strong immunogenicity. Therefore, it becomes an ideal candidate for personalized treatment using neoantigen cancer vaccines.
The clinical trial registration number of this study is NCT02316457, utilizing the TNBC-MERIT (mutational group engineered RNA immunotherapy) clinical treatment protocol. The study was conducted through a research strategy involving individualized vaccine preparation, clinical vaccination, multidimensional immune detection, and long-term clinical follow-up.
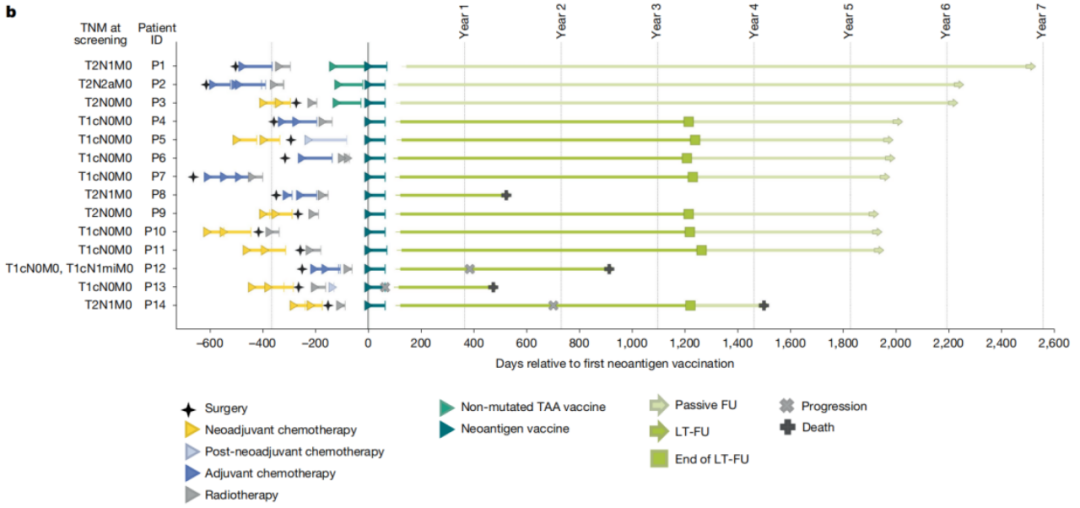
The study recruited a total of 15 patients with early-stage invasive TNBC confirmed by histology. The inclusion criteria were as follows: patients who had completed standard neoadjuvant/ adjuvant therapy within 1 year after surgery, with at least 5 neoantigens identifiable in the tumor, and no tumor recurrence. Among them, 14 patients completed the treatment regimen and were included in the final treatment efficacy evaluation.
The preparation process of individualized mRNA cancer vaccines is distinctly different from that of preventive pathogen vaccines such as COVID-19 vaccines. Researchers first perform gene sequencing on the tumor tissues and peripheral blood mononuclear cells of patients, and predict new tumor antigens capable of inducing T cell immunity using bioinformatics algorithms. Then, they assemble the selected tumor neoantigen gene sequences with secretory signal peptides that enhance T cell antigen presentation, as well as human leukocyte antigen I-like molecule transport domains (MITD). Finally, the mRNA is packaged in liposomal nanoparticles to construct an individualized cancer vaccine suitable for each patient. The average duration of this preparation process is 69days (34~125days), and each patient requires individual preparation for their own use.
The vaccination method for cancer vaccines is also different from that of preventive vaccines. Preventive vaccines are mostly administered subcutaneously, intradermally, or orally, while therapeutic cancer vaccines need to be implemented in conjunction with the patient's clinical treatment plan: patients first undergo intervention treatments such as surgery, chemotherapy, or radiotherapy, and then receive individualized mRNAcancer vaccines via intravenous injection. In this study, patients received a total of 8 doses of individualized vaccination, with the first 6 doses administered weekly, and the subsequent 2 doses administered every 2 weeksThe 1agent was completed on day 64. Among the 14 patients, 3 patients were first vaccinated with a bridging vaccine based on tumor-associated antigens (TAA), followed by a dose escalation to verify safety, involving 14.4μg to 50μg for the second dose. After safety assessment, the remaining 11 patients were directly vaccinated with the therapeutic target dose of 50μg.
In interventional clinical trials, patient safety is the primary evaluation indicator. In this study, the vast majority of treatment-related adverse events occurring in patients after vaccination were of grade 1 or grade 2, such as fever, headache, chills, and fatigue. These symptoms mostly appeared 1 to 3 days after vaccination and usually resolved spontaneously within 1 day. Only 1 patient discontinued the medication after receiving 3 doses due to adverse events such as grade 3 hypotension and grade 2 nausea. This result indicates that the overall safety of using this cancer vaccine is good. The antitumor effect of cancer vaccines is primarily achieved by activating the function of T cells in patients' bodies. Therefore, researchers employ methods such as IFN-γ enzyme-linked immunosorbent spot (ELISpot) assay and T cell receptor (TCR) sequencing to assess the immune status of patients after vaccination, with a focus on detecting the immune function of T cells. The results showed that the individualized mRNAcancer vaccine could induce a potent, multi-epitope neoantigen-specific Tcell response in patients: all 14patients induced a Tcell response to the vaccine neoantigens, with 9patients responding to at least 5neoantigens, and 86%of patients could detect neoantigen-specific Tcell responses through in vitro ELISpot detection; The vaccine-induced Tcell response is primarily de novo. Before vaccination, 41mutational targets showed no detectable Tcell response. After vaccination, a clear detection was observed, with only 2neoantigens exhibiting baseline T cell response, which was further amplified by the vaccine.
The study also found that the vaccine-induced Tcell response is long-lasting and the activated Tcell phenotype and function are diverse. Although the vaccine-induced multi-neoantigen Tcell response experiences a transient contraction after vaccination, it can still maintain a high level for 1 to 3.5 years; even without vaccine booster shots, some patients' neoantigen-specific CD8+Tcells can still be detected in peripheral blood after 6years post-vaccination. Vaccine-induced CD8+ T cells primarily consist of effector memory T cells and terminally differentiated effector memory T cell populations. Additionally, there exists a subpopulation of stem-like memory cell phenotype T cells, particularly in P1 patients. This group of cells co-expresses TCF-1 and IL-7Rα stem-like memory T cells, which can still be detected in peripheral blood after 6 years of vaccination.
▶▶How effective is it?
So, how effective is this cancer vaccine in treatment? The median follow-up period of this trial was up to 5years (62months, with a range of 15~80months). Among 14patients, 11showed no signs of recurrence; among them, P8died of unknown causes 15months after the last dose of vaccine, with no tumor recurrence before death. This high benefit result confirms the effectiveness of individualized mRNAcancer vaccine in the treatment of TNBC.
Although this trial achieved good therapeutic effects, it also exposed the complexity and challenges of tumor treatment: 3patients experienced disease recurrence during treatment. Among them, the P14patient, who had the weakest Tcell response induced by the vaccine, achieved complete remission after receiving anti-PD-1treatment, but subsequently developed systemic recurrence and died; P13On the surface of the primary tumor cells of patients, the expression levels of human leukocyte Iantigen molecules related to CD8+ Tcell response are low, and these molecules are almost completely absent in recurrent tumors. The expression of key genes in the antigen presentation pathway (such as B2M, HLAfamily genes) is downregulated. Even if there is functional Tcell infiltration induced by vaccines in patients, the tumor still achieves immune escape through antigen presentation barriers; P12
In summary, this study systematically explored the preparation of individualized neoantigen mRNA cancer vaccines, the immunization of tumor patients, and analyzed its safety, in vivo specificity Tcell response level, and therapeutic effect. Research has confirmed that this type of therapeutic personalized neoantigen mRNA vaccine possesses strong immunogenicity and can activate a durable CD8+ Tcell immune response in early-stage TNBCpatients after undergoing surgery and neoadjuvant /adjuvant therapy. It also demonstrates good clinical feasibility, controllable adverse reactions, and high safety. Simultaneously, the study has found that some patients may experience reduced vaccine efficacy due to immune escape mechanisms such as differences in tumor antigen immunogenicity and antigen presentation dysfunction. This provides valuable insights for optimizing vaccine treatment strategies in the future.