About Lehe
乐于心,和与众,与己乐,与人和; 心宽念纯,百病无生。

If you cannot understand the subsequent professional description, you can finish reading this text in two minutes
Overview of the condition
Knowledge
Lung Cancer Gene Testing
The identification of a series of oncogenic driver genes in lung cancer has further refined the classification of lung cancer from a purely histopathological classification to a molecular classification based on driver genes. EGFR is commonly found in lung adenocarcinoma.
The EGFR gene is a type of gene that expresses epidermal growth factor receptors, promoting normal cell division and proliferation under physiological conditions. However, in some cases, mutations in proto-oncogenes can lead to the development of tumors, with a higher incidence in non-smoking female lung adenocarcinoma. When it was discovered that EGFR mutations often occur in lung adenocarcinoma tumors, people began to consider how to inhibit tumor growth by targeting these mutations. Later, EGFR monoclonal antibodies, commonly known as "targeted drugs," were developed. Typically, EGFR mutations make lung adenocarcinoma more sensitive to specific targeted drugs.
After reviewing Ms. Chen's medical records, Professor Zhang Minghui made the following analysis and judgment:
1. The patient was diagnosed with early-stage lung cancer through a physical examination and underwent radical surgery. The postoperative staging was early. However, the patient has multiple nodules in the lungs, with risk factors such as ground glass and solid small nodules, which cannot be taken lightly.
2. The patient has not undergone genetic testing, and it is recommended to undergo genetic testing to guide subsequent treatment based on the results.
3. NKT therapy uses powerful immune cells to kill tumor cells that may remain undetected in the body, and has minimal side effects. It can be combined with other treatment options such as targeted drugs.
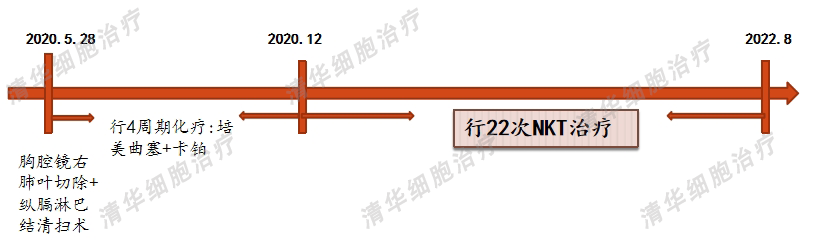
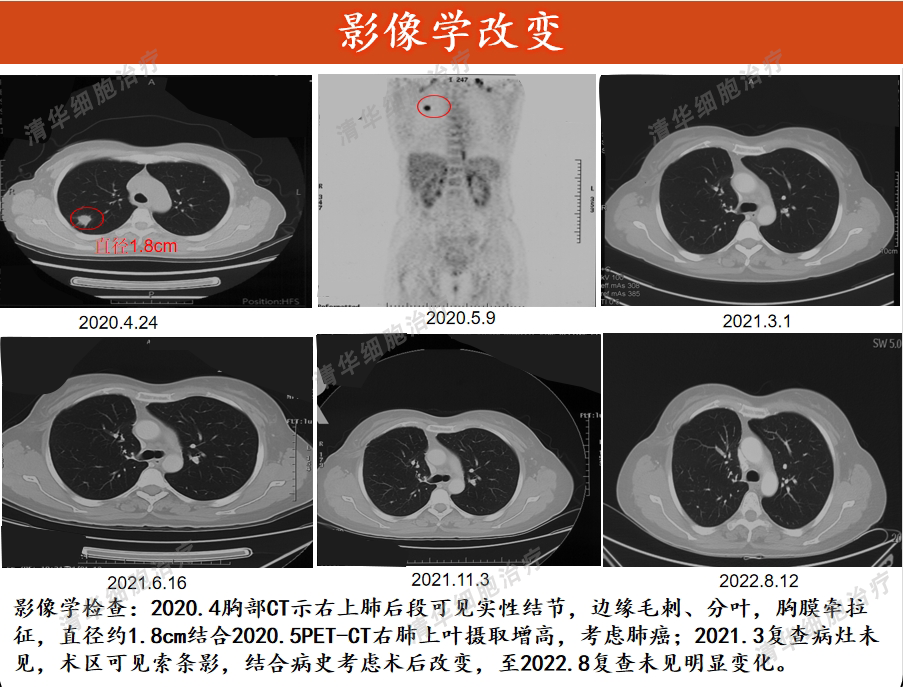
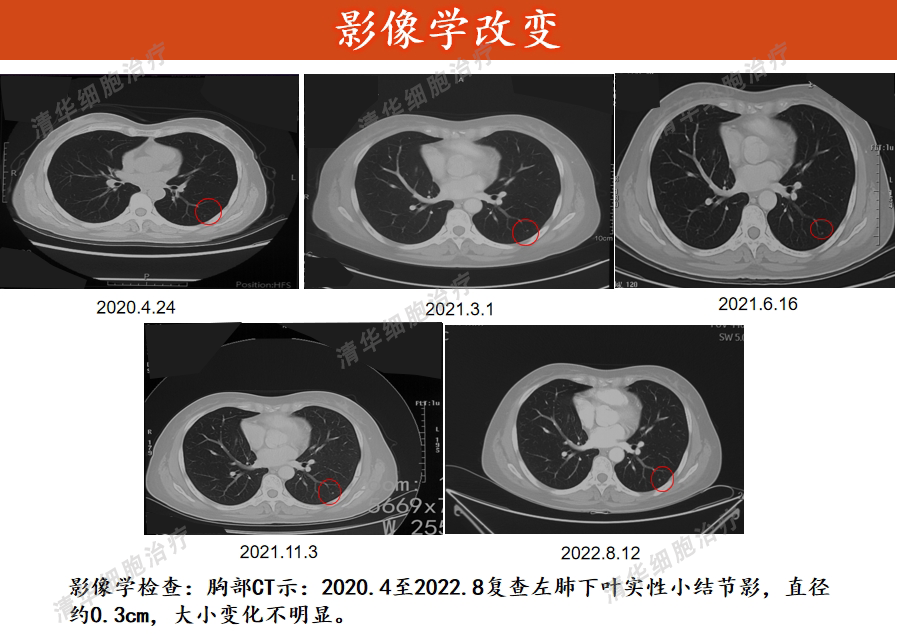
Image Aspects
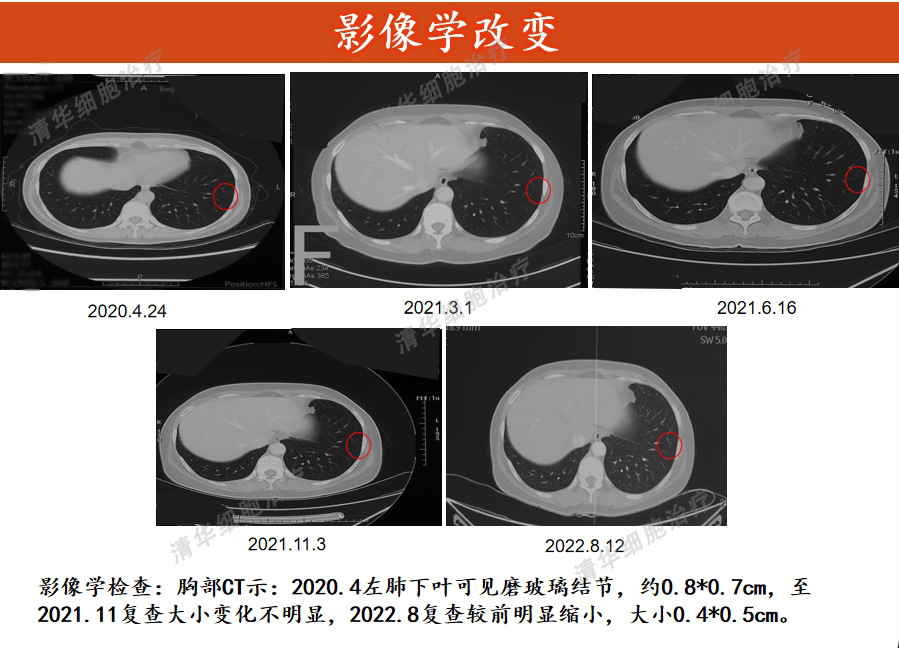
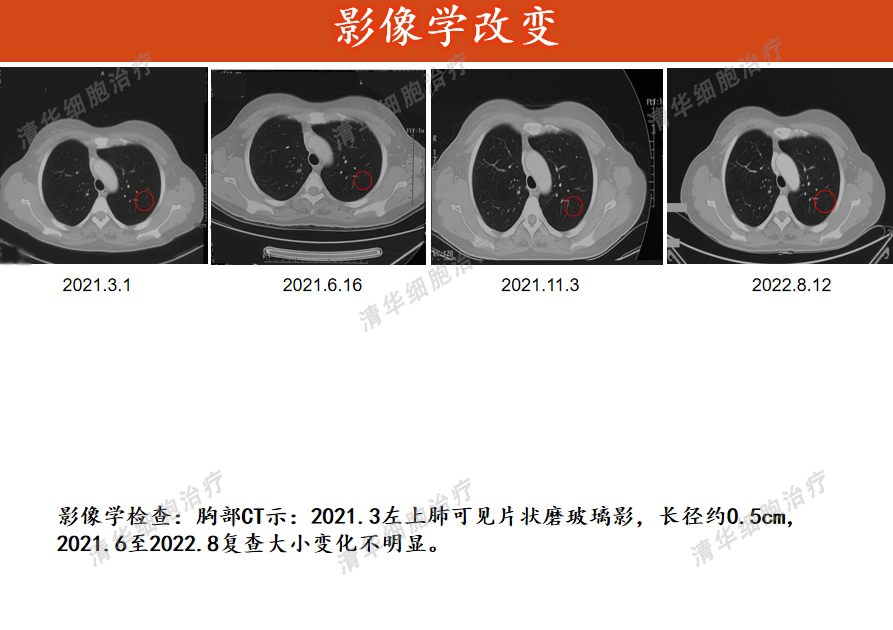
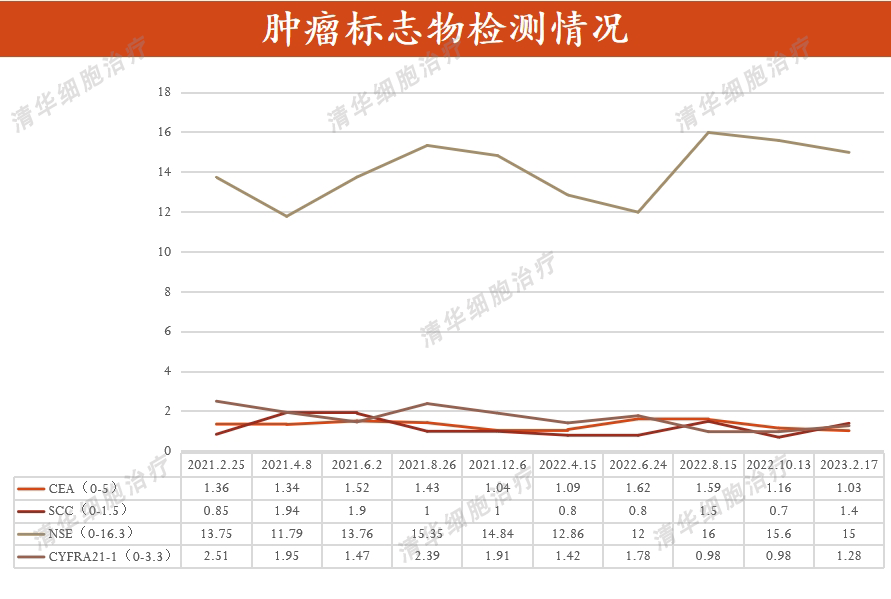
Tumor markers
Conclusion and Comments
Ms. Chen achieved the expected results after undergoing NKT cell therapy. After a long-term follow-up, there was no recurrence or metastasis of the primary tumor, and the pulmonary nodules remained stable without signs of canceration. Additionally, her quality of life improved.
Based on tumor volume, patients with stage Ia lung cancer are categorized into three groups: tumor volume ≤3cm³, 3-8cm³, and >8cm³. The five-year disease-free survival (DFS) rates for these groups are 88.0%, 73.6%, and 62.1%, respectively. This indicates that tumor volume is an independent risk factor for DFS and overall survival (OS) in stage I non-small cell lung cancer (NSCLC) after complete tumor resection.
Although Ms. Chen underwent surgery for stage I lung cancer, she still couldn't be complacent. To achieve long-term stability, she opted for NKT cell therapy and achieved the desired results. In this process, NKT cell immunotherapy played an indispensable role in reducing the risk of recurrence and metastasis, while strengthening the immune system, thereby ensuring long-term stability for patients.
Reference:< H362>
【1】Department of Thoracic Surgery, Sun Yat Sen University Cancer Center, China; State Key Laboratory of Oncology in Southern China and Collaborative Innovation Center for Cancer Medicine, Guangzhou, China; Lung Cancer Institute, Sun Yat Sen University, Guangzhou, China.
Click on the image